Image Credit: Nature
Research on obesity and related diseases is always a top demanding area in science. Although we all know the golden rule to reduce fat, which is working out or eating less, how this fat is actually reduced or what drives the motion is still unclear. But now a study published in the Nature journal has shed a light on this matter. It demonstrates that giving the ‘right’ signal to the neurons embedded in fat could make them break it down!
The right signal to do this is a hormone called ‘Leptin’ which is secreted by the fat itself. The researchers observed that, in the absence of leptin these nerves shrink. And they grew back when leptin is administered. More importantly, these fluctuations affected the stored fat to breakdown in mice.
Leptin and the magic it does
Secreted by the fat tissue, Leptin works as a control for energy reserves in fat tissue and regulate the brain to adjust food intake or appetite. This regulates the body weight. Mutations in the Leptin gene is directly linked with severe obesity, defected thermogenesis and disrupted fat burn in the body. Thermogenesis is where the body’s brown fat gets breakdown and creates heat without shivering. In this study, it was revealed that administering Leptin as a drug can reverse these issues.
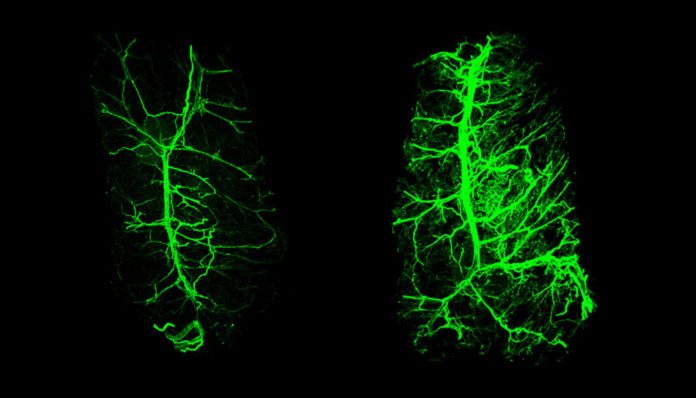
Researchers genetically engineered mice not to produce Leptin. Then they grew heavier, ate more and moved less. They also could not tolerate cold temperatures where other normal mice could, as their thermogenesis was off. And their fat tissue innervation was significantly diminished. This was same for mice who were resistance to Leptin. But this was reversed significantly by administering Leptin as a drug. They moved more, ate less, and within 2 weeks, the white fat began to breakdown producing more energy and they could gradually use the body’s ‘brown fat’ to generate heat. And the thermogenesis was back on! Sparse nerves when they were obese began growing back (Image). Thanks to Leptin, the regrowth of nerves was significantly connected to fat breakdown and boosted energy expenditure.
According to the researchers, “While the architecture of the nervous system can change significantly as a young animal develops, we did not expect to find this profound level of neural plasticity in an adult,”
Using novel imaging techniques, the researchers traced Leptin’s work from fat tissue to the brain’s hypothalamus and from there, a message came back through the spinal cord to the nerves in the white fat. It was believed to be the message of re-growth. It is the first study to prove Leptin’s ability to control the nerve growth embedded in fat, the white fat itself and also the brown fat. In other words, fat itself is controlling the presence of nerves in it and its function.
More studies are on the horizon to clearly determine the role of this pathway. Finding ways to reverse the resistance of Leptin in obese people is one such ambition. This will potentially open new ways to treat obesity and related metabolic disorders based on these nerves embedded in fat.