Credit: Gerd Altmann
A team of scientists have discovered that stem cells can regenerate nerves to support old damaged nerves and repair their motor functions in Parkinson’s disease. Although their study was based on a mouse model with Parkinson’s, it gives a solid direction as to how stem cell treatment could tackle this unfortunate degenerative disease.
Parkinson’s disease
The disease is caused by loss of nerves in a specific brain region called, ‘substantia nigra’. This causes dopamine, a chemical responsible for controlling movements of the body to be reduced. The reduction of dopamine is the main cause of many symptoms in Parkinson’s disease.
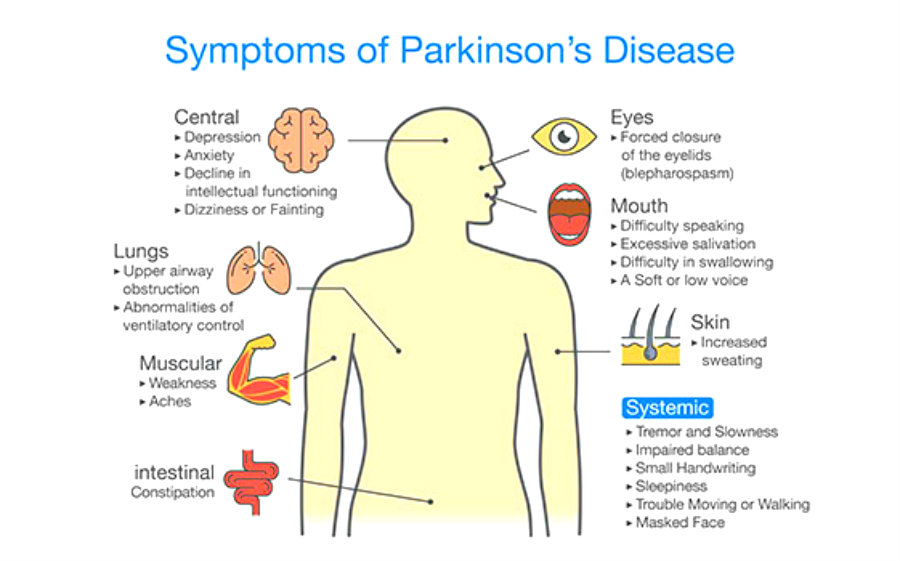
The statistics show that 1 in every 500 people are affected by this worldwide. And the symptoms usually begins when a person is over 50. There are treatments to reduce the severity of symptoms as the world is still waiting for a cure for Parkinson’s disease.
Explaining the basis of their study, “Our brain is wired in such an accurate way by very specialized nerve cells in particular locations so we can engage in all our complex behaviours. This all depends on circuits that are wired by specific cell types. Neurological injuries usually affect specific brain regions or specific cell types, disrupting circuits. In order to treat those diseases, we have to restore these circuits,” says Prof Zhang, at UW–Madison’s Waisman Center.
The study
First, the researchers made human embryonic stem cells to become dopamine-inducing neurons before implanting them into the mouse brain’s substantia nigra. And gave it another few months to be well established in the brain. The whole process had to be done carefully by identifying specific neurons that are important in doing the job.
The newly generated stem cell-induced nerves had nicely integrated and resulted in good motor functions in the mice with Parkinson’s. These new nerves had grown long distances to integrate with the motor control area of the brain. Not only that, but they also had connected with regulatory regions in the brain which stops them from overstimulating.
All these newly made connections allowed the implanted nerves to act just like the native nerves that produced dopamine.
How did they know it worked?
They had to make sure the stem cells they implanted were actually involved in the mouse’s improved motor skills. To do that, they added ‘an on and off switch’ into the stem cells which could be activated through specialised designer drugs given by food or injections.
So, the researchers noticed that when the stem cells were ‘switched off’ the mouse’s motor skill improvements were just gone, confirming the direct involvement of the stem cells to its newly gained motor skills. The researchers also believe that their ‘on/ off switch method’ could be used for many future applications to fine-tune transplanted cells. Interesting!
The study has been a result of many years of attempts. The team had been researching on stem cells’ ability to convert into different forms of neurons that would pave the way to find treatments for many neurological disorders. But it is easily said than done as each disease is connected with specific sets of neurons and identifying, targeting or selecting them is crucial and difficult before trying to generate them through stem cells.
Prof Zhang himself faced an accident that left him partially paralysed. And he himself was searching for a solution where stem cells could regrow specialised neurons that could help him and many others suffering from neurological disorders. Adding more hope to their new discovery, Prof Zhang says, “We used Parkinson’s as a model, but the principle is the same for many different neurological disorders. There is hope, but we need to take things one step at a time.”
And we wish them all the best!
The research study has now been published in the journal, Cell Stem Cell.